Introduction:
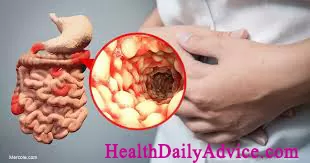
Crohn’s Disease, now more frequently referred to as Crohn’s disease, is an inflammatory bowel disease that, well, causes inflammation of the bowel. Unlike its cousin, ulcerative colitis, which only affects the large intestine, Crohn’s disease causes inflammation and tissue consumption anywhere along the gastrointestinal tract, from the opening to the anus.
Although ulcerative colitis is classified and treated as an autoimmune disease, Crohn’s disease isn’t technically classified as an autoimmune disease, but rather an immune-related disorder.
What does that mean exactly?
Well, with autoimmune, we think that your cells and proteins trigger the immune system to start attacking itself. In Crohn’s disease, the immune system is believed to be triggered by some foreign pathogen in the gastrointestinal tract.
Several pathogens have been implicated, like Mycobacterium par tuberculosis as well as Pseudomonas and Listeria species. So the immune systems react to foreign pathogens. Isn’t that
What it’s Assumed To Be Doing?
Well, Yes and No;
- Yes because it’s targeting a foreign invader,
- No, because the inflammatory response is large and uncontrolled and leads to the destruction of the cells in the gastrointestinal tract.
- So what is thought to happen is one of these pathogens activates the immune system by antigen presentation, meaning one of the gastrointestinal cells is like here, I think this is an infectious molecule, and that is fine because it is.
- At that point, T helper cells, or Th1 cells swoop in and release cytokines which are cell signaling molecules like interferon-gamma, and tumor necrosis factor-alpha, which further stimulates the inflammatory response.
- The cytokines attract Inflammatory cells like macrophages which start releasing, even more, inflammatory substances like proteases, platelet-activating factors, and free radicals, all of which contribute to inflammation.
- Although not definitively understood, it’s thought that for patients with Crohn’s disease, one of the steps in this process is dysfunctional and leads to an unregulated and out-of-control inflammatory response. Unregulated inflammation means lots of proteases, platelet-activating factors, and free radicals floating around the gastrointestinal tissue which ultimately destroys healthy tissue.
- This dysfunctional immune response is thought to be a product of genetics, and in fact, patients with family members who have Crohn’s disease are much more likely to develop it themselves. Some genes have been identified and are thought to contribute to the development of the disease.
- One of these is a frameshift mutation in the NOD2 gene, now called CARD15. Usually, for gene expression, nucleotides are read in groups of three. But if you add or subtract one or two nucleotides, it essentially shifts all the remaining nucleotides, usually ends up in totally different amino acids equaling coded, and probably a dysfunctional protein.
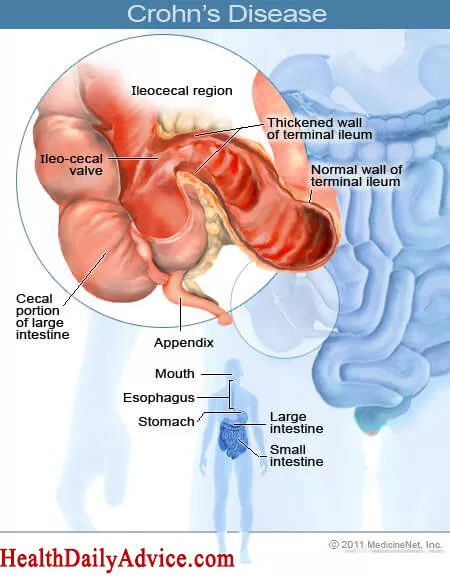
- Okay, so the intestinal wall, like that one, has much of ridges and grooves and also can be separated into the mucosal layer, submucosal layer, muscle layer, and serosa. Typically, pathogens aren’t supposed to be able to just pop right through the wall, but it’s thought that perhaps some defect in the epithelial barrier in Crohn’s disease more easily lets pathogens through.
- Once through, the immune system is activated through antigen presentation, which ultimately leads to the apparent unregulated inflammation just described, as cells become inflamed and destroyed, the immune cells invade down into the under mucosa, and organize themselves into granulomas, This guy over here is a granuloma, these are large masses of immune cells that are trying to encapsulate whatever they think is foreign.
- Eventually, from inflammatory and cellular damage, ulcers form, which are almost like craters in the intestinal wall. Unlike what we see in the other inflammatory bowel disease, like ulcerative colitis which extends through the mucosa and submucosa, the damage and inflammation in Crohn’s disease extend beyond the submucosal layer and through the whole depth of the intestinal wall, sometimes regarded as being transmural, from mucosa to serosa.
- Also, another distinction from ulcerative colitis is that these areas of inflammation are scattered as opposed to being continuous, and have areas of healthy tissue interspersed between inflamed areas, this gives it a cobblestone appearance.
- A final difference, as mentioned already, is that Crohn’s disease can affect any area of the GI tract, most commonly it affects both the ileum and colon, second just the ileum, and third just the colon, but again, those are just the most commonly seen, and it can affect any part of the GI tract, from the mouth to the anus.
Symptoms:
As more gastrointestinal tissue is damaged, patients might experience pain in the affected areas, most commonly in the right lower quadrant which is associated with the ileum. Also, diarrhea and blood in the stool are common symptoms.
Causes:
The blood is from damaged intestinal wall tissue, and if the cells in the large intestine are damaged, they lose their capacity to reabsorb water, and more water is excreted, leading to diarrhea. Along the same lines, if the small intestine is the one taking the brunt of the damage, patients may have male absorption issues, since the small intestines are super important for absorbing nutrients.
Treatment:
- Treatment for Crohn’s disease usually involves anti-inflammatory medications and antibiotics. Antibiotics are thought to help control symptoms by reducing bacteria levels in your intestine, which is sometimes called bacterial overgrowth, and also by curbing the immune response since some antibiotics have anti-inflammatory effects as well.
- If patients have severe symptoms, they may be given immunosuppressants like corticosteroids. Beyond that, surgery and removal of the affected tissue are possible, but one fundamental distinction from ulcerative colitis is that removal doesn’t cure the disease since inflammation can occur anywhere along the GI tract.
FAQS
Common symptoms include abdominal pain, diarrhea, weight loss, fatigue, and inflammation in various parts of the digestive tract. Individuals may also experience fever, rectal bleeding, and reduced appetite.
The exact cause of Crohn’s Disease is unclear, but it is believed to involve a combination of genetic, environmental, and immune system factors. Family history and certain environmental triggers may contribute to its development.
Diagnosis involves a combination of medical history, physical examination, imaging tests (such as endoscopy or colonoscopy), blood tests, and sometimes biopsies. These help confirm the presence of inflammation and rule out other conditions.
Treatment aims to control inflammation, relieve symptoms, and improve quality of life. Medications, such as anti-inflammatory drugs, immunosuppressants, and biologics, are commonly used. In severe cases, surgery may be necessary to remove damaged portions of the digestive tract.
Yes, certain lifestyle modifications can complement medical treatments. These may include dietary changes, stress management, regular exercise, and avoiding tobacco. However, individual responses to lifestyle changes can vary, and it’s crucial to consult with healthcare professionals for personalized advice.
Note:
So in This Post, Crohn’s Disease – Symptoms, Causes and Treatment What other points can you think of/have experienced? Let me know in the comments.
If you found this helpful or feel free to share your experience if you can relate to these points and if you are comfortable share
For More Articles Related to Crohn’s Disease Stay Tuned To our Site









